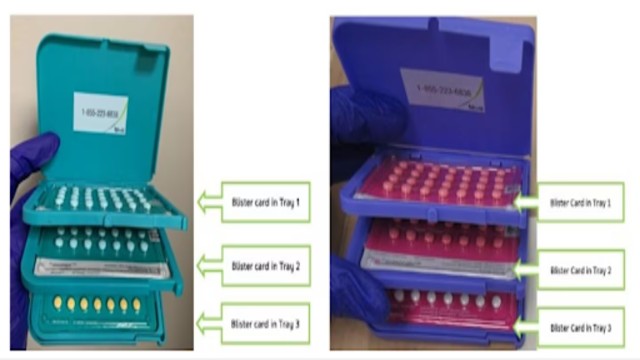
Midodrine tablets (left) for low blood pressure and MAR-Amlodipine tablets (right) for high blood pressure are seen in this composite photo.
A nationwide blood pressure medication recall has prompted Health Canada to urge patients to check their prescriptions carefully.
The alert follows the discovery of a medication mix-up involving a commonly prescribed treatment for high blood pressure.
Officials say the issue could pose serious health risks if it goes unnoticed.
Health Canada confirmed that two specific lots of MAR-Amlodipine 5 mg tablets are affected.
Some bottles may contain the wrong medication, creating potential danger for patients managing hypertension.
What Triggered the Blood Pressure Medication Recall
MAR-Amlodipine is widely prescribed to treat high blood pressure and chest pain.
However, the manufacturer, Marcan Pharmaceuticals Inc., reported an unexpected packaging error.
During production, tablets meant for treating low blood pressure were mistakenly placed in some MAR-Amlodipine bottles.
As a result, patients expecting one medication may have received another with opposite effects.
Health Canada classified the situation as serious due to the possible consequences of incorrect dosing.
How to Identify the Correct Medication
Patients are being advised to inspect their medication immediately.
The correct MAR-Amlodipine tablets have a distinct appearance that is easy to recognize.
They are white to off-white, eight-sided tablets with the numbers “210” and “5” printed clearly.
The incorrectly packaged tablets are midodrine 2.5 mg.
These tablets are round and marked with “M2,” making them visibly different.
Midodrine is prescribed to treat low blood pressure, not high blood pressure.
Why the Mix-Up Is Dangerous
Health Canada warns that accidentally taking midodrine instead of amlodipine can be harmful.
Midodrine raises blood pressure, which can be dangerous for patients already managing hypertension.
Potential side effects include sudden spikes in blood pressure, dizziness, and fainting.
In severe cases, the error could lead to organ damage if not addressed promptly.
The risk increases if patients continue taking the wrong medication without realizing the mistake.
What Patients Should Do Immediately
Anyone who finds round or unfamiliar tablets in a MAR-Amlodipine bottle should stop using them.
Health Canada advises returning the medication to the pharmacy right away.
Pharmacists can provide guidance and ensure patients receive the correct prescription.
Patients are also encouraged to speak with their healthcare provider if they have concerns.
Checking medications regularly is especially important during a blood pressure medication recall.
When to Seek Medical Help
If someone has already taken the wrong medication, medical advice should be sought quickly.
Health Canada recommends contacting a healthcare professional if unusual symptoms appear.
Emergency services should be called if severe dizziness, slow heartbeat, or very high blood pressure occurs.
Immediate medical attention is also critical for chest pain or sudden, severe headaches.
Symptoms such as slurred speech, weakness, or numbness require urgent evaluation.
Ongoing Monitoring and Awareness
Health Canada continues to monitor the situation closely.
The agency stresses the importance of patient awareness and medication safety.
Recalls like this highlight why patients should review prescriptions before use.
For now, officials urge Canadians to remain cautious and informed.
Prompt action can prevent serious harm and ensure safe blood pressure treatment continues uninterrupted.