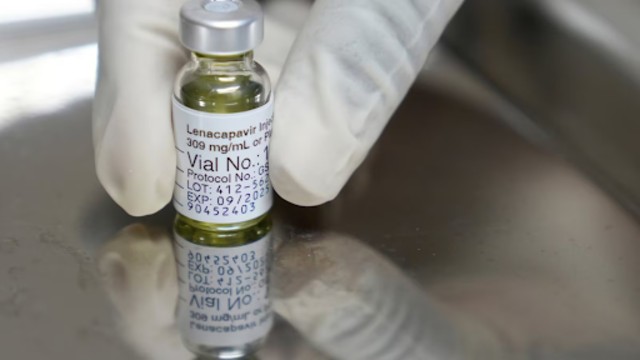
A pharmacist holds a vial of lenacapavir, an injectable HIV prevention drug, at the Desmond Tutu Health Foundation's Masiphumelele Research Site, in Cape Town, South Africa, Tuesday, July 23, 2024, which was one of the sites for Gilead's lenacapavir drug trial. (AP Photo)
A breakthrough in HIV prevention has just made history. The U.S. Food and Drug Administration (FDA) has approved the world’s first twice-a-year shot to prevent HIV. Developed by Gilead Sciences, this long-acting injectable could change how we approach HIV prevention forever.
A New Hope in the HIV Fight
Called lenacapavir, this drug offers protection for six months with just one injection. It showed near-perfect results in clinical trials, drastically outperforming the daily pills currently used for PrEP (pre-exposure prophylaxis). Some health experts believe this could become the most powerful tool yet in the battle against HIV.
Although not a vaccine, lenacapavir works as a potent shield against the virus. The twice-yearly dose could attract people who struggle with daily pills or face stigma for taking them.
“This could truly end HIV transmission,” said Greg Millett from amfAR, The Foundation for AIDS Research.
From Daily Pills to Twice-Yearly Convenience
Condoms still play a role, but modern prevention efforts increasingly rely on PrEP medications. Lenacapavir, now branded as Yeztugo for prevention, stands out. It offers the longest protection of any available option — administered only twice a year.
The injection is given under the skin in the abdomen and slowly releases the drug over time. It’s already sold under the name Sunlenca for treating HIV, but this new use focuses solely on prevention.
Access Hurdles Could Dim the Promise
Despite the excitement, challenges remain. The U.S. is dealing with health system cutbacks and reduced funding for global HIV initiatives. These gaps, according to Millett, make it difficult to ensure people even return for the two yearly doses.
Gilead hasn’t revealed the drug’s price yet. And importantly, the shot only prevents HIV — not other sexually transmitted infections.
Global HIV Goals in Jeopardy
Worldwide, hopes to end HIV by 2030 are fading. In the U.S. alone, over 30,000 people are newly infected each year. Globally, that number exceeds 1.3 million. Just 400,000 Americans currently use any form of PrEP, despite millions being at risk.
Studies show high PrEP usage can reduce infections. But access remains uneven. Many new cases involve women — often needing protection they can use privately.
One large study in South Africa and Uganda gave lenacapavir to over 5,000 women and girls. Not a single one contracted HIV. In contrast, about 2% of those taking daily pills became infected. A second study among gay and gender-diverse individuals showed similar success.
A New Era of Prevention
Houston’s Ian Haddock is among the early users. He struggled with daily PrEP routines but found freedom with lenacapavir. “Now I don’t even think about it,” he said.
The drug simplifies protection. Just two clinic visits a year. No daily pill. No judgment.
Experts say the next step is ensuring access for everyone at risk — everywhere.