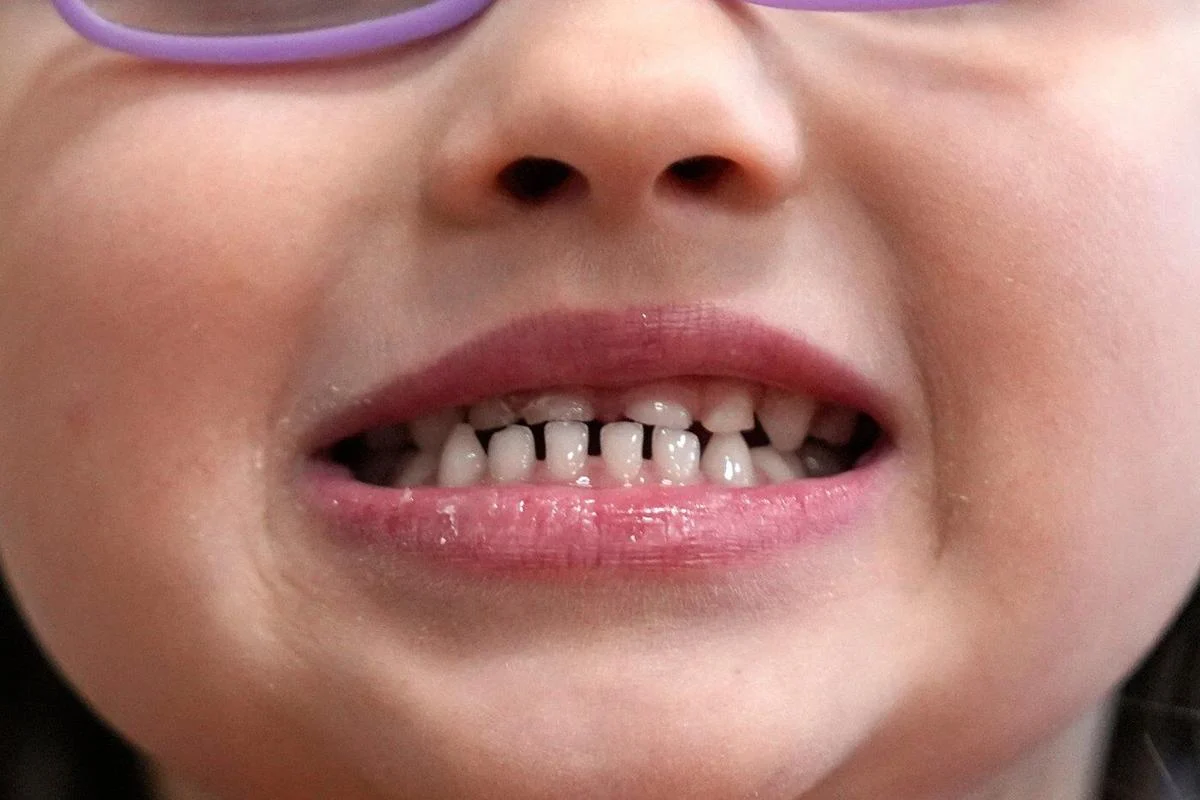
A child shows off her teeth after a dental exam in Concord, N.H., Wednesday, Feb. 21, 2024. (AP Photo)
The U.S. Food and Drug Administration (FDA) has announced new restrictions on fluoride supplements for children, citing potential health risks linked to their use. The decision marks a significant shift in federal policy regarding a chemical long considered vital to dental health.
The agency said the products should no longer be used for children under three years old or for older children who are not at serious risk of tooth decay. Until now, fluoride drops and tablets were commonly prescribed for children as young as six months.
Cautious Step, Not a Complete Ban
Friday’s move stops short of an earlier indication by the FDA that the products might be removed entirely from the market. Instead, the agency issued warning letters to four companies, instructing them not to promote their fluoride supplements outside the new safety limits.
Fluoride tablets, lozenges, and drops are typically recommended for children in areas where drinking water lacks adequate fluoride levels. These supplements are meant to strengthen enamel and reduce the risk of cavities, especially in communities without fluoridated water.
New Findings Raise Questions
Alongside the policy update, the FDA released a new scientific review suggesting that the benefits of fluoride supplements for children are modest and that their use may pose emerging health risks.
The analysis linked fluoride intake to potential issues such as gut disturbances, weight gain, and cognitive changes.
“For the same reason fluoride can fight bacteria on teeth, it may also affect the gut microbiome, which could have broader health effects,” the FDA explained in its statement.
Dentists and healthcare providers were also sent advisory letters urging caution and awareness about these risks.
Divided Opinions Within the Dental Community
The American Dental Association (ADA) has strongly disagreed with the FDA’s stance, maintaining that fluoride supplements are safe when prescribed properly. The organization says there is no solid evidence linking recommended fluoride levels to serious health problems.
While the FDA acknowledged that excess fluoride can cause tooth discoloration or white spotting, it emphasized growing scientific concern over its possible systemic impacts.
Rural Dentists Fear More Cavities
Dental experts warn the restrictions could lead to a rise in cavities, particularly in rural or low-income regions where water is not fluoridated. Many of these communities rely heavily on fluoride supplements to maintain oral health in children.
Meanwhile, Health Secretary Robert F. Kennedy Jr., who has long criticized fluoride use, continues to advocate ending the practice of adding it to public water supplies nationwide. Kennedy, a former environmental lawyer, has called fluoride a “dangerous neurotoxin” associated with developmental and neurological risks.
What the New Rule Doesn’t Change
The FDA clarified that its latest decision applies only to dietary fluoride supplements such as drops, tablets, and lozenges. Fluoride-based toothpastes, mouthwashes, and in-office dental treatments remain unaffected.
The Centers for Disease Control and Prevention (CDC) continues to support water fluoridation, saying it strengthens teeth by replacing lost minerals and remains one of the most effective tools in preventing tooth decay.
Still, Friday’s announcement signals a growing shift in how U.S. health authorities approach fluoride exposure — a debate that now extends far beyond the dentist’s chair.