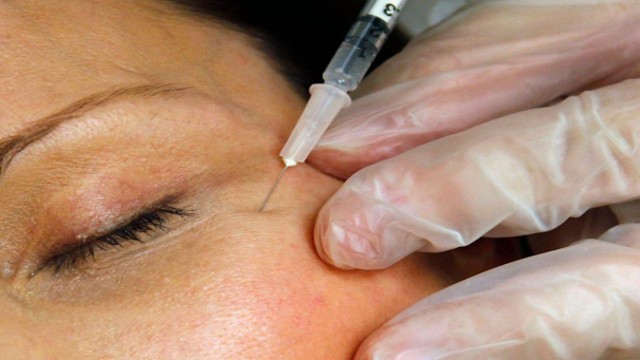
A patient receives a Botox injection at a clinic in Arlington, Va., on June 5, 2009. (AP Photo)
U.S. health authorities have issued stern warnings to multiple online sellers accused of distributing unapproved and counterfeit versions of Botox and similar injectable wrinkle treatments.
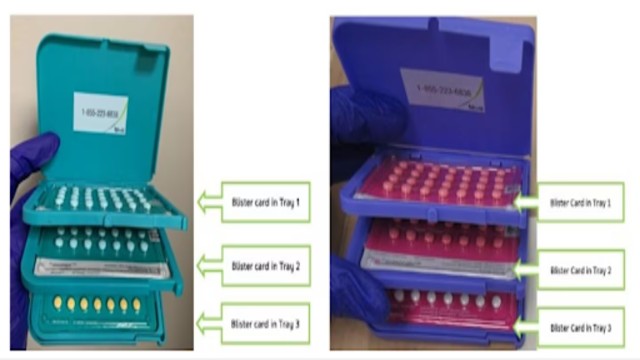
The Food and Drug Administration (FDA) confirmed that warning letters were sent to 18 websites found selling illegal or misbranded products for cosmetic use. The action follows reports of injuries and toxic side effects linked to these unverified injectables.
Reports of Harm Spark Investigation
According to the FDA, several consumers have suffered harmful reactions after using these unapproved Botox-like products. Some experienced symptoms consistent with botulism — a serious condition that can affect muscle control and breathing.
Botox, a purified and diluted form of botulinum toxin, is among the most potent biological substances known. In regulated doses, it works by blocking nerve signals and relaxing targeted muscles. The FDA approves Botox not only for smoothing facial wrinkles but also for treating medical conditions like migraines, eye disorders, and muscle spasms.
FDA Targets Cosmetic Retailers
Most of the websites flagged by the FDA were beauty and cosmetic sellers marketing injectable products that mimicked Botox. The agency stated that the items were falsely labeled or contained ingredients not approved for human use.
“Unapproved and counterfeit injectables pose serious health risks,” the agency emphasized. “Consumers should not purchase or use products claiming to be Botox unless obtained directly from a licensed medical provider.”
The FDA reminded the public that only Botox and its FDA-approved alternatives — including those cleared since Allergan first introduced the product in 1989 — meet safety and quality standards for human use.
Serious Health Risks Linked to Fake Injectables
Botox and similar botulinum-based medications carry a boxed warning, the FDA’s strictest alert, due to potential life-threatening side effects. When misused or improperly formulated, the toxin can spread beyond the injection site, weakening or paralyzing muscles essential for breathing and swallowing.
Symptoms of botulism may appear hours after injection and include difficulty breathing, slurred speech, muscle weakness, or trouble swallowing.
FDA Urges Public to Seek Licensed Care
In its official statement, the FDA urged patients to seek only trained, licensed healthcare professionals for injectable treatments. The agency also advised anyone experiencing symptoms of botulism after injection to seek immediate medical attention.
“The safety of consumers is our top priority,” the FDA said. “We will continue to monitor online marketplaces and take action against those who distribute unsafe or unapproved medical products.”
Growing Concern Over Online Beauty Sales
Experts warn that the rising popularity of online beauty treatments has made it easier for counterfeit medical products to circulate. Many unauthorized sellers operate internationally, making it difficult to trace product origins or verify ingredients.
The FDA’s latest move highlights the growing dangers of buying cosmetic injectables online — a reminder that trusted results begin only with certified professionals and approved products.