Infants Get Their First Malaria-Specific Treatment. The Canadian Press
In a historic decision, Swissmedic—Switzerland’s medical regulator—has approved the first-ever malaria drug made just for infants. The new medicine, Coartem Baby, developed by Swiss pharmaceutical giant Novartis, is designed for babies weighing between 2 and 5 kilograms (roughly 4.5 to 11 pounds).
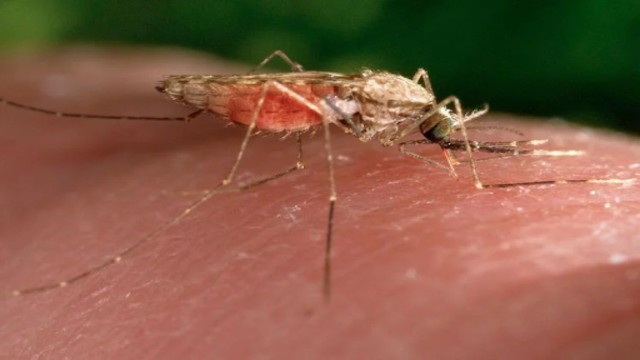
It's a major step in addressing malaria’s deadly impact, particularly in Africa, where most of the world’s malaria deaths occur. In 2023 alone, over 597,000 people died from malaria—95% of them in Africa. Tragically, more than three-quarters of these victims were children.
First Malaria-Specific Treatment for the Most Vulnerable
Until now, babies with malaria were treated using medicines intended for older children. These drugs had to be carefully adjusted to avoid overdose—a risky, less-than-ideal workaround that many health professionals called “suboptimal.”
Coartem Baby changes that. It’s a lower-dose version of an existing treatment, made specifically for newborns. It combines two antimalarial agents in one pill, ensuring a safer, easier, and more effective treatment option for the infants.
“This is a drug we know works and is safe,” said Dr. Quique Bassat, director-general at the Barcelona Institute for Global Health. “Very young and underweight children need properly dosed treatments, not makeshift solutions.”
Fast-Tracked for Urgent Impact
Swissmedic fast-tracked the approval of Coartem Baby under a rare special authorization, supported by the World Health Organization (WHO). This accelerated process is designed to speed up access to critical medicines in developing nations.
Only two other treatments have been granted this type of approval by Swissmedic in the past—underlining how urgently this medicine is needed.
Eight African Nations Next in Line
Eight African countries were part of the medicine’s assessment and are expected to approve the drug within the next 90 days. Novartis has announced plans to distribute Coartem Baby in malaria-endemic regions “largely not for profit.”
But experts are urging the company to be more specific about that claim.
Calls for Transparency and Fair Access
Dr. Bhargavi Rao, co-director at the Malaria Centre of the London School of Hygiene and Tropical Medicine, applauded the move but pushed for clarity.
“We need transparent pricing, clear timelines, and information on which countries will benefit,” she said. “Without that, it’s hard to measure real impact.”
Dr. Rao also pointed out that despite recent efforts—including new vaccines and mosquito control programs—malaria cases continue to rise in parts of Africa, especially in crisis-stricken areas.
A Timely Breakthrough Amid Global Setbacks
This milestone comes at a time when resistance to current malaria drugs is growing and international funding programs are shrinking. Health advocates hope Coartem Baby will reignite support and deliver life-saving impact where it’s needed most.
Though it took over two decades since the original Coartem was approved for older age groups, this infant-specific version marks a new chapter.
It’s a long-awaited lifeline for families in malaria-prone regions—and a reminder that age-specific solutions can no longer be overlooked in global health.