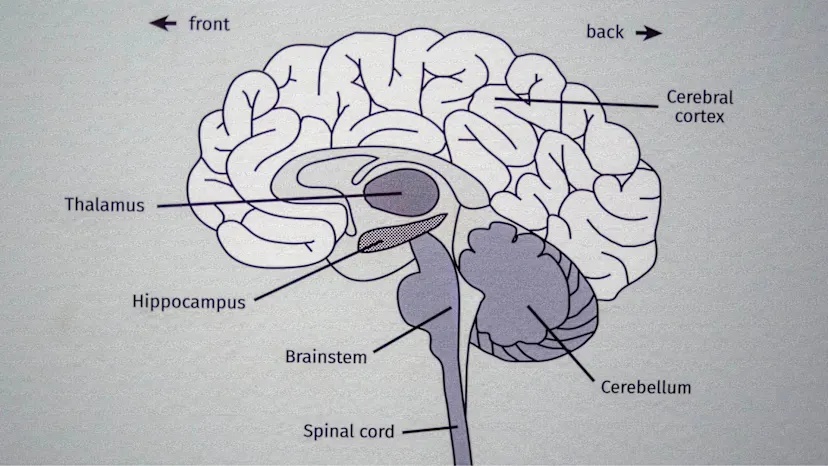
Two brain scans side-by-side. The one on the left is healthy, the one on the right shows loss of brain matter as neurons die in Huntington's disease. Credit: UCLH
Breakthrough Gene Therapy Offers Hope Against Huntington’s Disease
Doctors have announced a historic breakthrough in treating Huntington’s disease, one of the most devastating inherited conditions. For the first time, the progression of the illness has been dramatically slowed through an experimental gene therapy.
Huntington’s disease, which destroys brain cells, carries symptoms resembling dementia, Parkinson’s, and motor neurone disease combined. Patients often begin to show signs in their 30s or 40s, and the illness is typically fatal within two decades.
Professors Ed Wild and Sarah Tabrizi led the UK part of the trial. Credit BBC
A Remarkable Slowdown
Results from a recent trial revealed that the treatment slowed the disease’s progression by an average of 75 percent. This means patients who might have experienced a year of decline could now expect four years of similar stability.
Professor Sarah Tabrizi, director of the University College London Huntington’s Disease Centre, described the findings as “spectacular.” She told media that such a result was beyond what researchers ever imagined.
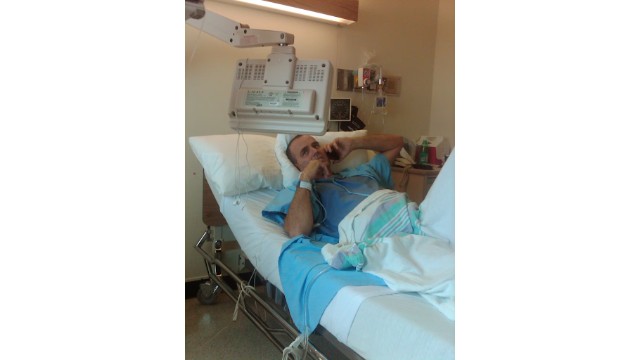
One patient, who had been medically retired, has returned to work following the treatment. Others expected to require wheelchairs are still able to walk.
The Science Behind the Breakthrough
The therapy involves an advanced form of gene treatment, delivered during a 12–18 hour brain surgery. Surgeons use real-time MRI scanning to guide a microcatheter into two key brain regions.
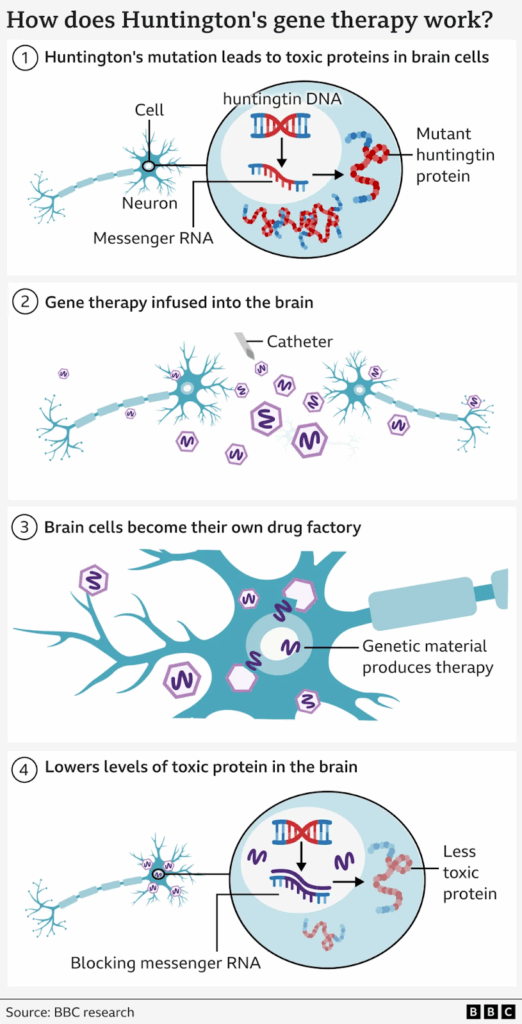
A modified virus, acting as a carrier, then delivers a new sequence of DNA directly into brain cells. These cells begin producing small fragments of genetic material designed to block harmful instructions that create toxic huntingtin protein.
This single-dose treatment lowers the levels of the mutant protein, which normally kills neurons. Early results also show reduced signs of brain cell death in spinal fluid samples.
Families See a Brighter Future
For families living under the shadow of Huntington’s, the news has brought an emotional wave of relief.
Jack May-Davis, a 30-year-old barrister’s clerk, inherited the faulty gene from his father, Fred, who died in 2016 at the age of 54. Jack had always expected to share the same fate, until now.
“This is absolutely incredible,” he said. “It allows me to think my life could be longer. The future feels brighter.”
Professor Ed Wild, consultant neurologist at University College London Hospitals, admitted the results brought him to tears. “We weren’t sure we would ever see this outcome,” he said. “The scale of impact is breathtaking. It’s hard to capture what this means for families.”
Jack May-Davis has the gene that causes Huntington's disease
Safety and Challenges Ahead
While the treatment has shown remarkable safety, some patients experienced inflammation from the viral delivery, leading to headaches and confusion. Most cases resolved naturally or with steroids.
Experts believe the therapy could last a lifetime, as brain cells do not renew themselves in the same way as skin or blood.
However, the cost remains uncertain. Gene therapies are notoriously expensive, with some treatments priced in millions per patient. Still, researchers argue that the long-term benefits could make it affordable in the bigger picture.
UniQure, the company behind the therapy, plans to seek U.S. approval in early 2026, with launches expected later that year. Discussions with European and UK regulators are also planned.
A young Jack with his father, Fred, who died of Huntington's disease.
A New Dawn for Huntington’s
Around 75,000 people across the UK, U.S., and Europe are currently living with Huntington’s, while hundreds of thousands more carry the gene and face a likely diagnosis.
Professor Tabrizi called this trial “the beginning.” She praised the volunteers as “truly brave” and said her team is now exploring whether early treatment in symptom-free carriers could delay or even prevent the disease altogether.
Dr. Walid Abi-Saab, chief medical officer at uniQure, echoed the optimism. He described the therapy as having the potential to “fundamentally transform” the future of Huntington’s disease.
For a condition long regarded as unstoppable, this breakthrough marks the first real sign of hope. Families once resigned to decline and loss can now imagine years — even decades — of better quality life.