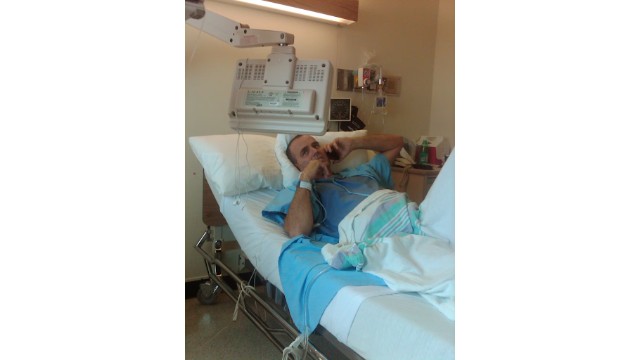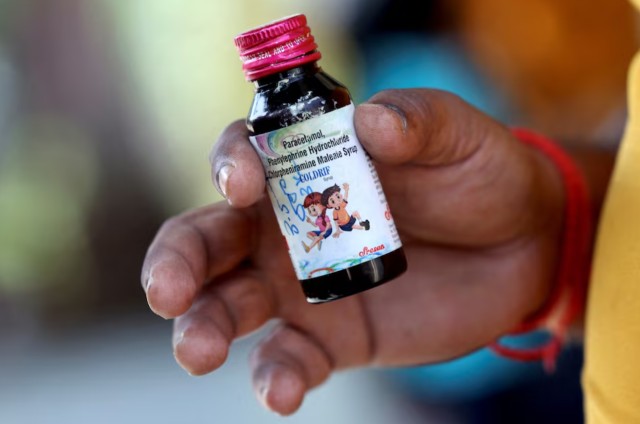
Nilesh Suryavanshi, 32, the father of a 3 year old child who has been admitted at the Government Medical College, holds a bottle of Coldrif cough syrup, which has been linked to the deaths of 17 children, in Nagpur, India, October 8, 2025. Photo Credit: REUTERS
Seventeen children under the age of five have died in India after consuming cough syrups contaminated with toxic chemicals, prompting global concern and renewed scrutiny of the country’s drug safety system.
Authorities confirmed that the deaths occurred over the past month after the children consumed a cough medicine containing diethylene glycol (DEG) — a poisonous compound found at levels nearly 500 times above the safe limit. The deaths were linked to the locally sold Coldrif syrup, which was banned after lab tests confirmed contamination on October 2.
Two More Cough Syrups Under Scrutiny
Following the Coldrif ban, authorities in Gujarat and other states issued a public warning against two more cough syrups — Respifresh and RELIFE — after tests also detected the same toxic chemical. The alert described diethylene glycol as “a highly dangerous substance” capable of causing severe poisoning, kidney failure, neurological damage, and even death, especially among children.
Officials have assured that the contaminated syrups were sold only within India. However, the World Health Organization (WHO) expressed concern that some products could have reached other countries through unofficial channels.
WHO Raises Alarm Over “Regulatory Gap”
The WHO confirmed it had been informed by Indian authorities about the three tainted syrups but noted that the source of contamination remains unclear. In a statement, the agency said it was “deeply concerned” and highlighted “a regulatory gap in screening for diethylene glycol and ethylene glycol in medicines sold domestically.”
Since 2023, India has required cough syrup exports to undergo government lab testing following the deaths of over 140 children in Gambia, Uzbekistan, and Cameroon linked to similar contamination. However, medicines sold only within India have not faced the same level of scrutiny.
Factory Under Investigation
The Coldrif syrup was manufactured by Sresan Pharmaceutical in Tamil Nadu. Police have launched a manslaughter investigation against the company, and its manufacturing license may soon be revoked.
When inspectors visited the plant earlier this week, they found the facility shut. Notices were pasted on its walls seeking details of raw materials and production processes. Burnt medicines and empty syrup bottles were seen scattered around, releasing a sharp chemical odor.
Inspections Widen Across States
India’s Drug Controller General, Rajeev Raghuvanshi, said nationwide inspections uncovered “serious lapses” at several drug factories, including failures to test every batch of ingredients as required by law. The health ministry confirmed that 19 manufacturing units across six states are now under inspection.
Among them are Shape Pharma and Rednex Pharmaceuticals, producers of RELIFE and Respifresh. Samples from both companies were declared “not of standard quality,” and their operations have been halted pending further investigation.
Global Concern Over India’s Pharma Safety
The recurring cases of toxic syrups have damaged India’s reputation as a leading global supplier of affordable medicines. The country’s $50 billion pharmaceutical industry ranks third in volume worldwide, after the U.S. and China.
India supplies around 40% of generic drugs used in the United States and over 90% of medicines distributed across several African nations.
As investigations continue, the WHO has reiterated its global advisory against the use of cough and cold medicines for children, urging strict vigilance and transparency in drug manufacturing.